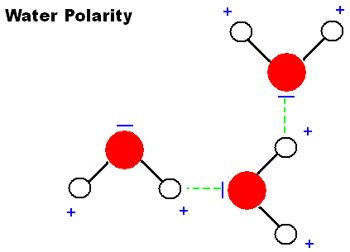
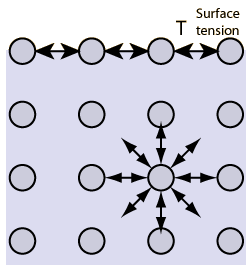
The Properties Of Water.
polarity: The Polar molecule makes one end positive and the other end negative.
cohesion: The tendency for water molecules form weak bonds and they stick to each other.
adhesion: The tendency of water to stick to other substances.
capillary action: Is a combination force of attraction among water molecules and all the surrounding materials causes the liquid to go up against the force of gravity.
surface tension: Is a force that acts on the the particles at the surface of a liquid.
specific heat:Is the amount of the heat needed to increase the temperature.
hydrogen bond: is the electromanetic attractive interaction between polarmolecules.
1. Circle the letter of each sentence that is true about water's structure
. a. Water is made up of atoms bonded to form molecules.
b. Water contains half as many hydrogen atoms as oxygen atoms.
c. Water molecules tend to push away from each other.
d. The chemical formula for water is H20.
2. One side of the water molecule has a positive charge, while the other side has a negative charge. What do the charges indicate about the molecule?
a. Water is a polar molecule.
c. Water is an ionic compound.
b. Water is a nonpolar molecule.
d. Water is an ion.
3. Bonds that form between water molecules are called Hydrogen bonds.
4. True or False? Hydrogen bonds are strong and require a lot of energy to break. False
5. The tendency for water molecules to stick to other water molecules is called Cohesion.
6. A force that acts on the particles of a liquid at the surface is called a Surface Tension
7. How does surface tension force the surface of water to curve? Gravity and Water forces this.
8. The tendency for water molecules to be attracted and stick to other substances is called a Adhesion
9. Circle the letter of each sentence that is true about water's surface tension.
a. It helps some insects "skate" across the surface of the water.
b. It refers to the tightness across the surface of the water.
c. It is caused by polar molecules repelling each other.
d. It causes raindrops to form round beads.
10. What is a curved surface in a graduated cylinder called? A meniscus.
11. Circle the letter of each sentence that is true about capillary action.
a. It explains how water moves against the force of gravity.
b. It is due to the attraction among molecules of water and surrounding materials.
c.It prevents water from moving through materials with pores.
d. It causes clothing to stay dry.
12. How does capillary action allow water to climb up the sides of a straw?
Capillary action is the combined force of attraction among water
molecules and with the molecules of surrounding materials causing a liquid to climb upward against the force of gravity.
13. A mixture that forms when one substance dissolves another is called a(n) Solution
The substance that does the dissolving is called a(n) Solvent
14. Why can water dissolve many substances?
Water is polar. Meaning that it is able to dissolve substances that have either positive or negative charge.
15. Circle the letter of each substance that dissolves in water
a. salt
b. oil
c. oxygen
d. wax
16. A substance that hates, or “fears” water is called Hydrophobic
17. The amount of heat needed to increase the temperature of a certain mass of a substance by 1°C is its Specific Heat
18. True or False? Compared with other substances, water requires a lot of heat to increase its temperature. True
19. Circle the letter of each sentence that is true about water's specific heat.
a. It is due to the many attractions among water molecules
. b. It makes large bodies of water heat up more quickly than nearby land.
c. It makes large bodies of water cool off more slowly than nearby land.
d. It leads to warmer air over land than over water on summer days.
20. How does the high specific heat of water affect your body? It helps maintain the inside of your body.
Match the terms review.
1. cohesion 2. capillary action 3. adhesion _____4. polarity _____5. hydrogen bond _____6. surface tension 7. specific heat
a. tendency to stick to other substances
b. tightness caused by the pulling of water molecules on each other
c. the amount of heat needed to raise 1kg of a substance 1o
d. weak bonds formed between water molecules
e. tendency to stick to other water molecules
f. uneven distribution of charges in a molecule
g. tendency to climb up due to cohesion and adhesion properties of water
1.e 2.g 3.a 4.f 5.d 6.b 7,c